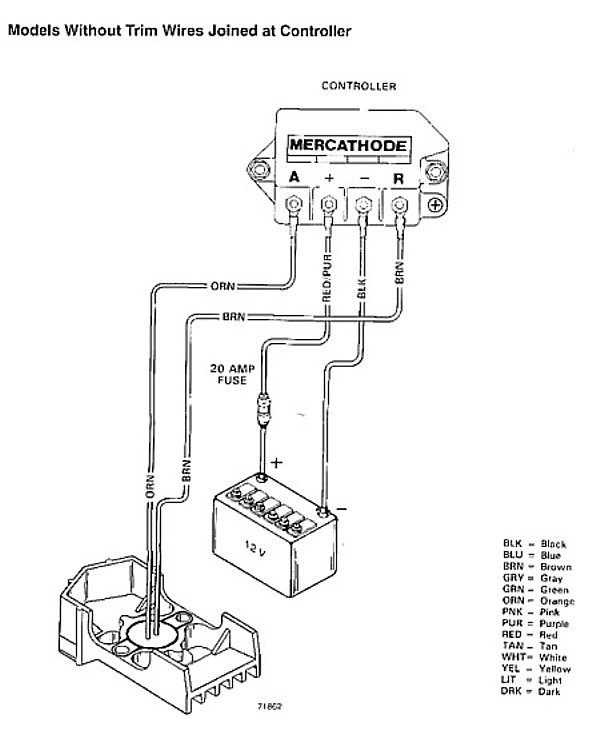
For example, reversing the current direction in a Daniell galvanic cell would produce an electrolytic cell, where the copper electrode is the positive terminal and the anode. In a recharging battery, or an electrolytic cell performing electrolysis, the cathode is the negative terminal, from which current exits the device and returns to the external generator. For example, the Daniell galvanic cell's copper electrode is the positive terminal and the cathode. It is continued externally by electrons moving inwards, this negative charge moving inwards constituting positive current flowing outwards. This outward current is carried internally by positive ions moving from the electrolyte to the positive cathode (chemical energy is responsible for this "uphill" motion). Look at this picture: BU-306: What is the Function of the Separator? Source Wikipedia: In a discharging battery or a galvanic cell, the cathode is the positive terminal since that is where the current flows out of the device (see drawing). Is this correct: "When charging, a buildup of electrons forms on the cathode"? I think it should be "When charging, a buildup of electrons forms on the anode". (See BU-306: Separator and BU-307: Electrolyte) The separator segregates the anode from the cathode, forming an isolator for electrons but allowing ions to pass through. In a flooded battery system, the electrolyte moves freely between the inserted electrodes in a sealed cell, the electrolyte is normally added to the separator in a moistened form.
Cathode anode diagram Activator#
Ion flow is made possible with an activator called the electrolyte. Table 2d: Composition of primary alkaline battery. Lithium ions move back to the positive electrode Metal oxides derived from cobalt, nickel, manganese, iron, aluminum Tables 2a, b, c and d summarize the composition of lead-, nickel- and lithium-based secondary batteries, including primary alkaline. The cathode of a battery is positive and the anode is negative. (See BU-212: Future Batteries) With few exceptions, lithium-metal batteries are non-rechargeable. Here the cathode is carbon and the anode metallic lithium. The anode of Li-ion is carbon (See BU-204: How do Lithium Batteries Work?) but the order is reversed with lithium-metal batteries. Since the battery is an electric storage device providing energy, the battery anode is always negative. A vacuum tube, diode or a battery on charge follows this order however taking power away from a battery on discharge turns the anode negative. This appears to violate the convention as the anode is the terminal into which current flows. The battery anode is always negative and the cathode positive. The electrode of a battery that releases electrons during discharge is called anode the electrode that absorbs the electrons is the cathode. The separator electrically isolates the electrodes but allows the movement of ions. Ions are atoms that have lost or gained electrons and have become electrically charged. On charge, the current flows in the other direction.Ī battery has two separate pathways one is the electric circuit through which electrons flow, feeding the load, and the other is the path where ions move between the electrodes though the separator that acts as an insulator for electrons. Release is by a passing current from the positive cathode through an external load and back to the negative anode. This leads electrons moving towards the cathode, creating a voltage potential between the cathode and the anode. When charging, a buildup of positive ions forms at cathode/electrolyte interface. An electrochemical battery consists of a cathode, an anode and electrolyte that act as a catalyst.
